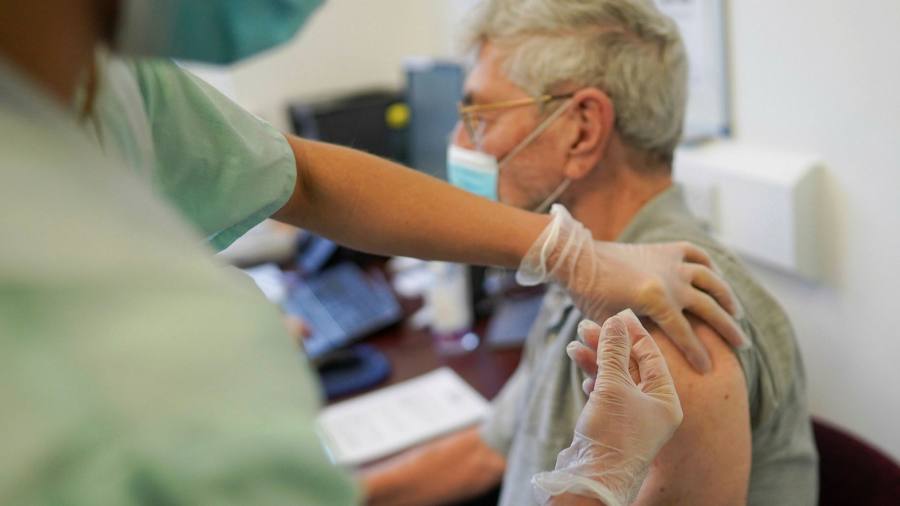
Pfizer criticized the UK’s decision to widen the gap between doses of Covid-19 vaccine, saying the “safety and efficacy” of the new schedule had not been evaluated as infections and hospitalizations continued to rise in the UK .
The rare intervention by the US pharmaceutical company comes amid growing controversy over the move, with doctors complaining about having to cancel appointments and reassuring anxious patients who were previously told they needed the second dose to keep them safe .
‘[Our] study. . . is designed to evaluate the safety and efficacy of the vaccine according to a two-dose schedule, separated by 21 days, ‘the company said.
“The safety and efficacy of the vaccine were not evaluated on different dosing schedules, as the majority of trial participants received the second dose within the window specified in the study design.”
The new dosing guidelines, approved by the UK Joint Committee on Vaccination and Vaccination and released on Wednesday, allow the second dose of both the vaccine developed by Pfizer with BioNTech and the newly approved Oxford / AstraZeneca vaccine to three applied. months after the first.
The change aims to give a first dose of vaccine to as many people as possible, in an effort to counteract the spiral numbers caused by a much more contagious viral variant of the coronavirus.
There was again evidence Thursday of the toll the virus is taking in the UK, with another 964 deaths announced and new signs of tension in hospitals.
NHS providers, representing health organizations across the UK, said pressure on Covid-19 was increasing, particularly in London and the south-east, where the increase in admissions over the past few days had been “extremely worrying”.
Saffron Cordery, deputy chief executive of NHS Providers, said there were 22,713 Covid-19 patients in hospital in England as of Wednesday – an increase of more than 27 per cent in seven days. “During the same period, the number of Covid-19 patients in critical care beds increased by 35 percent,” she added.
Matt Hancock, the UK’s health secretary, on Thursday tried to resist proposals that the vaccination program, which the government said was necessary to stem the pandemic, was behind schedule.
He said 944,539 people in the UK had their first dose of Covid-19 vaccine. ‘The NHS has a clear delivery plan and today’s figures show once again how our fantastic NHS has risen to this enormous task. . . Now that we have approved a second vaccine, we can expect this number to increase rapidly in the coming months. ”
Matt Hancock said more than 940,000 people had their first shot of a Covid-19 vaccine. © Jessica Taylor / UK Parliament
Although partial protection by the vaccine seems to start as early as twelve days after the first beat, Pfizer emphasized that two doses are needed to provide maximum protection against the disease, with an efficacy of 95 percent. It is important to say: “There is no evidence to show that protection is maintained after the first dose after 21 days.”
The drugmaker said that decisions on alternative dosing regimens were in the hands of health authorities and that he was committed to talking to regulators, but stressed that each recipient should receive the maximum possible protection, “which means immunization with two doses of the vaccine “.
The Regulatory Agency of Medicines and Healthcare Products stated: ‘The decision was taken to update the dose interval recommendations for the BioNTech / Pfizer vaccine after a thorough review of the data by the MHRA’s working group Covid-19 Vaccines Benefit Risk Expert. The expert group concluded that the efficacy of the vaccine would be maintained at dosing intervals of longer than 21 days. ”
Doctors in the UK expressed anger and concern over the switch, saying they had to spend time canceling appointments for people who were expecting to receive their supplemental dose over the next few days.
Helen Salisbury, a family doctor in Oxford, described the situation as trivial and calculated that it would take 193 hours for the network of practices to administer the vaccine to her environment to reschedule the appointments.
She questioned the judgment and data modeling behind the decision. “What does science say? We do not know. ”
The British Medical Association has said it is unreasonable and totally unfair to ask doctors to book appointments of tens of thousands of elderly and vulnerable patients, and to support practices that discuss existing appointments for the next few days.
In his New Year’s message, British Prime Minister Boris Johnson praised the Oxford vaccine, saying it ‘literally offers a new life to people in this country and around the world’.
However, he warned of “a tough battle that lies ahead for weeks and months as we face a new variant of the disease that requires a new vigilance”.
Four questions about the deployment of Oxford / AstraZeneca in the UK
Why is there a six-day delay between the approval of the regulation and the
first vaccinations with the Oxford / AZ vaccine?
Vaccines must be delivered and stored according to the conditions set by the British Medicines Regulator, the MHRA, which was first known with the approval announcement on Wednesday.
What other factors are delaying the implementation of the new vaccine?
Jonathan Van-Tam, deputy chief executive officer, said this week that “the only thing we are going to slow down is that quantities of vaccine are available”. According to him, it was not only about the production of vaccines, but also about ‘filling and finishing’ – the process of placing the vaccine in vials and packaging it for distribution – which he said was a critical short source about the whole world ‘was. The second factor that delays the supply for patients is the regulatory requirement that each group of vaccine be checked for safety and quality before it can be given to patients.
Will change the dosing regimen to one dose and a second thereafter
Speed up 12 weeks?
The decision to increase dose intervals, not only for the Oxford / AstraZeneca vaccine, but also for the BioNTech / Pfizer version, aims to protect people faster. Half of the stock does not have to hold back to give second doses to those who have already received some protection, but all the available doses can be used immediately. However, the government and the NHS will be wary of previous big promises of the pandemic that have not materialized about how much more will be covered as a result in a specific time scale.
When will the UK be able to reach the target of 2 million people?
week to be vaccinated?
The government stands by its promise that all those aged 50 and under whose health is particularly vulnerable will be vaccinated by the end of March. Boris Johnson promised “tens of millions of doses” on Wednesday. Simon Stevens, head of England’s NHS, spoke of everyone in the priority groups covered by ‘late spring’.